The Implementation of Technology Enabled Clinical Trials is Driving the Global Virtual Clinical Trials Market
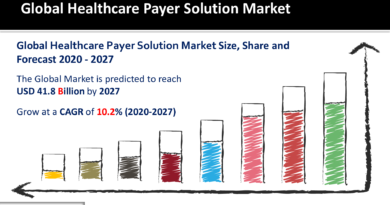
According to Ameco Research, the global virtual clinical trials market size is expected to grow a CAGR above 5% over the projected timeframe and reach a market value of around USD 17,466 Million by 2030.
Trials utilizing patient-facing technologies, such as smartphone apps, tablets, or wearable sensors, have been referred to by a number of different names. They are frequently referred to as virtual, decentralized, remote, direct-to-patient, and hybrid trials.
By enhancing patient experiences with the help of digital technologies as well as other remote services, virtual trials provide an original solution to these issues. Since 2014, a number of specialized start-ups have appeared in the field of virtual trials. These businesses provide all of the services required to carry out a virtual trial, such as patient recruitment, telemedicine, eConsent, in-home medical visits, data collection and analysis, remote monitoring device integration, eCOA collection, and DtP drug supply. Science 37 and Medable, which were established in 2014 and 2015, respectively, are leaders in this industry. Since the beginning of the pandemic, interest in these businesses has increased. 18 collaborations had already been established this year as of mid-June 2021.
Download Sample Pages Of this report – https://www.amecoresearch.com/sample/276665
Global Virtual Clinical Trials Market Growth Aspects
VCTs are an alteration of clinical trials that reduce costs, save time, and improve participant experience. They are not a new or distinct type of clinical trial. With the aid of digital health technologies, VCT is able to boost participant diversity and representation, improve retention, and recruit more quickly. The difficulties associated with traditional clinical trials, such as scheduling numerous lengthy appointments during the day, are also overcome by VCT.
The industry anticipates a global opportunity with the evolution of contract research organizations (CROs) into virtual trial providers. Before to the Covid-19 pandemic, few CROs provided a full variety of virtual clinical trial solutions to customers, though the trend toward these trials was well under way. Since the pandemic started, CROs have been very active in this area, especially through partnerships and mergers of home healthcare service providers, eClinical software providers, and remote patient monitoring (RPM) service providers to strengthen their virtual trial capabilities.
Although patient retention rates are still a concern, with a high percentage of participants leaving studies before they are finished, trial recruitment rates emerge to be improving. The key to enhancing the general trial experience and raising retention rates is to make trials more patient-centric. A patient-centric study must be planned and carried out with the needs of participants at its core, making sure that the patient voice is included. It has been demonstrated that involving patients in the design of the study protocol enhances the quality of the research, patient outcomes, and participant relevance, all of which can boost enrollment, adherence, and retention.
Sponsors can include a bigger population for this study thanks to virtual studies, which enhances enrollment, participation, and retention. Additionally, it makes it possible for digital health technologies to continuously collect real-time data. The effort, time dedication, and load on the applicants, CRCs, and investigators may eventually be significantly reduced by virtual connectivity, monitoring, and management.
Market Segmentation
The global virtual clinical trials market segmentation by Ameco Research based on study design and indication. Based on the study-design, the market is divided into interventional, expanded access, and observational. Among them, the interventional study-design generated significant virtual clinical trial market revenue. Based on the indication, the market separated into cardiovascular, oncology, and others. Among them, the oncology segment achieved a noteworthy virtual clinical trial market share in 2021 and is likely to continue its dominance during the forecast timeframe 2022 to 2030.
Regional Overview
North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa are the regional classification of the global virtual clinical trials market. Based on our virtual clinical trials market regional outlook, North America region gathered maximum amount of share during the projected years 2022 to 2030. This is due to increased government support, increased introduction of advanced technologies in clinical research, and increased R&D in this area. Meanwhile, the Asia-Pacific region is expected to register the fastest growth rate (CAGR) in the coming future. Some of the key factors supporting the Asia-Pacific virtual clinical trial market include increased availability of a huge patient pool enabling easy recruitment of candidates and increased penetration of digital technologies.
Major Players
Some key virtual clinical trials companies covered in the report are IQVIA, Covance Inc., ICON plc, Parexel International, Medable, Inc. PRA Health Sciences, Oracle Corporation, LEO Innovation Lab, CRF Health, Medidata Solutions, Clinical Ink, Inc.
To meet various trial requirements throughout the trial lifecycle, clinical trial solution providers offer a wide variety of decentralized solutions. Virtual clinical trials have advanced significantly over the past few years thanks to digitalization and technological advancements.
Buy this premium research report – https://www.amecoresearch.com/buy/276665
Would like to place an order or any question, please feel free to contact at sales@amecoresearch.com & Richard@amecoresearch.com
Contact No: + 1 407 915 4157